Botanical extracts are natural ingredients that contain diverse polyphenol type molecules. Some are well known for their endogenous and exogenous antioxidant abilities. However, when speaking under the feed and pet food regulatory framework of the European Union so far only one natural product is registered as an antioxidant, and this is mixed-tocopherols. Some botanical extracts are registered but classified as flavoring compounds.1 This article will focus on the exogenous antioxidant capacity of rosemary and olive extracts and the main reaction mechanism of their active molecules with the highest antioxidant potential.

Technical Service Manager
Kemin Nutrisurance EMEA
ROSEMARY EXTRACT
Even if rosemary extract is well known for its great antioxidant activity for controlling oxidation of fats, oils and fat rich matrices, in feed and pet food regulation it is still classified as a flavoring compound.1
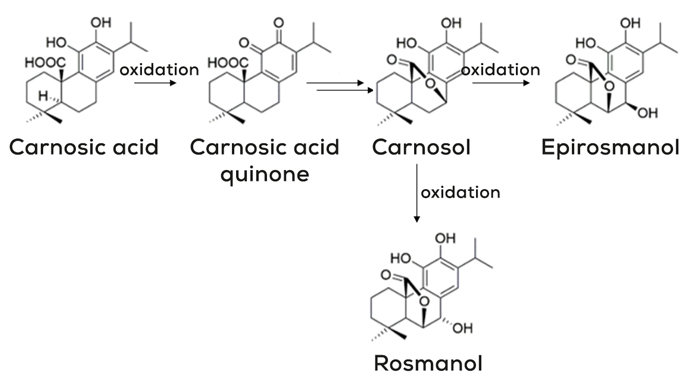
Rosemary extract contains different diterpene molecules with antioxidant activity, from these, the one with the highest antioxidant effect is carnosic acid. Other molecules such as carnosol, rosmanol or epirosmanol have also some antioxidant capacities but less significant than carnosic acid. The reason for that is that all the above are decomposition products of carnosic acid, as we will see while explaining the simplified chemical reaction mechanisms.
OXYGEN SCAVENGER MECHANISM
To prevent oxidation rosemary extract, or specifically, carnosic acid can scavenge singlet oxygen, an excited form of oxygen, and free radicals. While doing this carnosic acid will oxidize itself to form a carnosic acid quinone that by action of temperature or other catalysts will be transformed into carnosol. The carnosol molecule will further follow this path and will oxidise itself to be transformed into epirosmanol or rosmanol, depending on the stereochemistry of the reaction.2,3 See Figure 1.
HYDROGEN TRANSFER MECHANISM
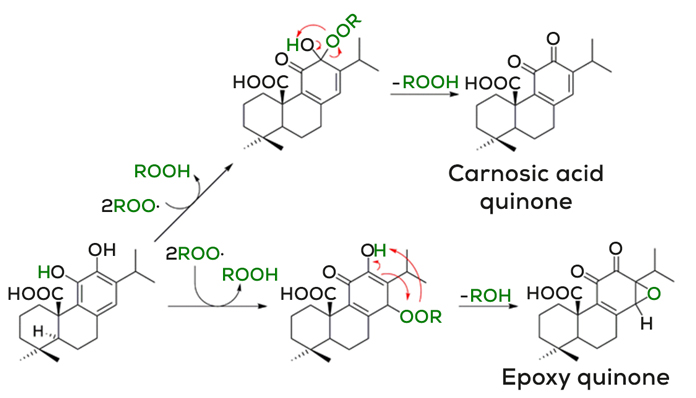
Carnosic acid can also neutralize free radicals by following a different mechanism, by donating hydrogen atoms to stabilize lipid molecules (in figure 2 represented as ROOH). This type of mechanism is called hydrogen transfer mechanism. Here carnosic acid can neutralize the free radical activity of two lipid radical molecules (ROO°) to form a carnosic acid quinone. Like in the previous mechanism the carnosic acid quinone can be transformed into carnosol to provide additional antioxidant activity. Or the mechanism can follow a different pathway and lipid radicals can be stabilized by forming an epoxy quinone. In summary, the key message here is that one molecule of carnosic acid can neutralize two lipid radical molecules.2 2
What we can learn from the chemistry explained here is that carnosic acid can generate a variety of secondary antioxidants. This unique cascade-type process is likely to amplify the antioxidative power of carnosic acid and hence of rosemary extract and to constitute an effective defense mechanism.3 In sum, the main driver for the antioxidant activity of rosemary extract is carnosic acid. This means the higher the carnosic acid concentration in a rosemary extract, the better will be its antioxidant activity. The rest of the phenolic diterpenes will also have some antioxidant activity but lower than carnosic acid because, as we have seen, they are degradation products from the carnosic acid molecule. Besides, rosemary extracts contain also other antioxidants such as rosmarinic acid bringing more antioxidant power to the overall activity of this botanical extract.
and hydroxytyrosol
OLIVE EXTRACT
Olive extract is another natural ingredient very well known for its health benefits and widely used for centuries in the countries of the Mediterranean area.
Apart from its health or endogenous antioxidant and antimicrobial properties, recent research has shown the potential benefit of using olive extract as an exogenous antioxidant to prevent oxidation of fats, oils and fat containing matrices.
Since olives are a raw material extensively used in human nutrition, the question arose if it could be economically suitable as an ingredient for the feed and pet food industries. Not all parts of the olive tree are used at the moment for human consumption. Among others, there are the leaves and there is the waste of the olive fruit after production of olive oil. Both co-products are rich in polyphenols with potential antioxidant activity.
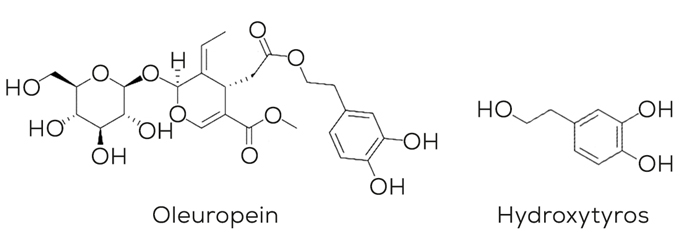
In olive materials we can find two interesting molecules for our antioxidant purpose: oleuropein and hydroxytyrosol. These two catechols have shown endogenous and exogenous free radical scavenging activity.4
Although we can find both active ingredients in olive leaf and fruit, olive leaves are rich in oleuropein while olive fruits main ingredient is hydroxytyrosol.
Oleuropein has shown an important antimicrobial effect as well as some endogenous antioxidant activity.5 Nowadays there are a number of supplements in the market for human but also for petfood claiming immune system boost thanks to the properties of oleuropein (just type on your web browser pet food supplement with oleuropein and you will find several companies offering such products). Unfortunately, as technological additive to control oxidation of fats and oils, oleuropein as such has shown a poor antioxidant activity in comparison with hydroxytyrosol and other known antioxidants.6
On the contrary, hydroxytyrosol has been demonstrated to be an ideal candidate to be considered as exogenous antioxidant to prevent the oxidation of feed and pet food relevant matrices.7,8

A few studies have proposed the mode of action of hydroxytyrosol, mainly by using radical scavenging reaction mechanism in which the hydroxytyrosol molecule can neutralized lipid radicals (ROO°). See figure 4.
Kemin internal results have shown that the antioxidant activity of olive fruit extract can be enhanced by combination with known natural antioxidant molecules and that contrary to rosemary extract and carnosic acid, higher amounts of hydroxytyrosol in an olive extract do not necessarily mean better antioxidant activity.9
Several studies have shown that olive fruit extract containing hydroxytyrosol but not only shows antioxidant activity comparable to that of pure hydroxytyrosol, perhaps due to the presence of active derivatives of hydroxytyrosol and other phenolic compounds which promote synergistic effects in the extract that are not available when using the pure molecule.
Further research is needed to elucidate the mode of action of the antioxidants present in the olive extract.
References:
1https://ec.europa.eu/food/food-feed-portal/screen/feed-additives/search accessed on 11th November 2023
2T. Masuda, Y. Inaba, Y. Takeda, J. Agric. Food Chem, 2001,49,5560-5565;; S. L. Richheimer, M. W. Bernart, G. A. King, M. C. Kent, D. T. Bailey, J. Am. Oil. Chem. Soc. 1996, 73, 507-514;
3M. Loussouarn, L. Krieger-Liszkay, A. Bily, S. Birtić, M. Havaux, Plant Physiol. 2017 Nov;175(3):1381-1394
4M. Fiorini, V. Crognaletti, O. Sabry, P. Fattori, Processes 2021, 9, 433.
5D. Borjan, M. Leitgeb, Molecules 2020, 25, 5946
6M.H. Gordon, F. Paiva-Martins, M. Almeida, J. Agric. Food Chem., 2001, 49, 2480-2485
7A.Y. V. Theah, T. O. Akanbi, Antioxidants 2023, 12, 929
8Martínez-Zamora, R. Peñalver, G. Ros, G. Nieto, Foods. 2021 Oct 28;10(11):2611
9Kemin R&D internal results
About Dr. Cristina Murcia García
Dr. Cristina Murcia García gained her PhD in Chemistry with focus on red-ox reactions from the University of Bonn (Germany). Since then, she has specialized in managing oxidation in rendering and pet food products. She works as Technical Service Manager for Kemin Nutrisurance Europe where she provides solutions to improve raw materials and pet food safety and shelf-life. Besides, she develops tailor-made trials and protocols for customers and holds customer specific trainings on topics like oxidation, freshness, and food safety.